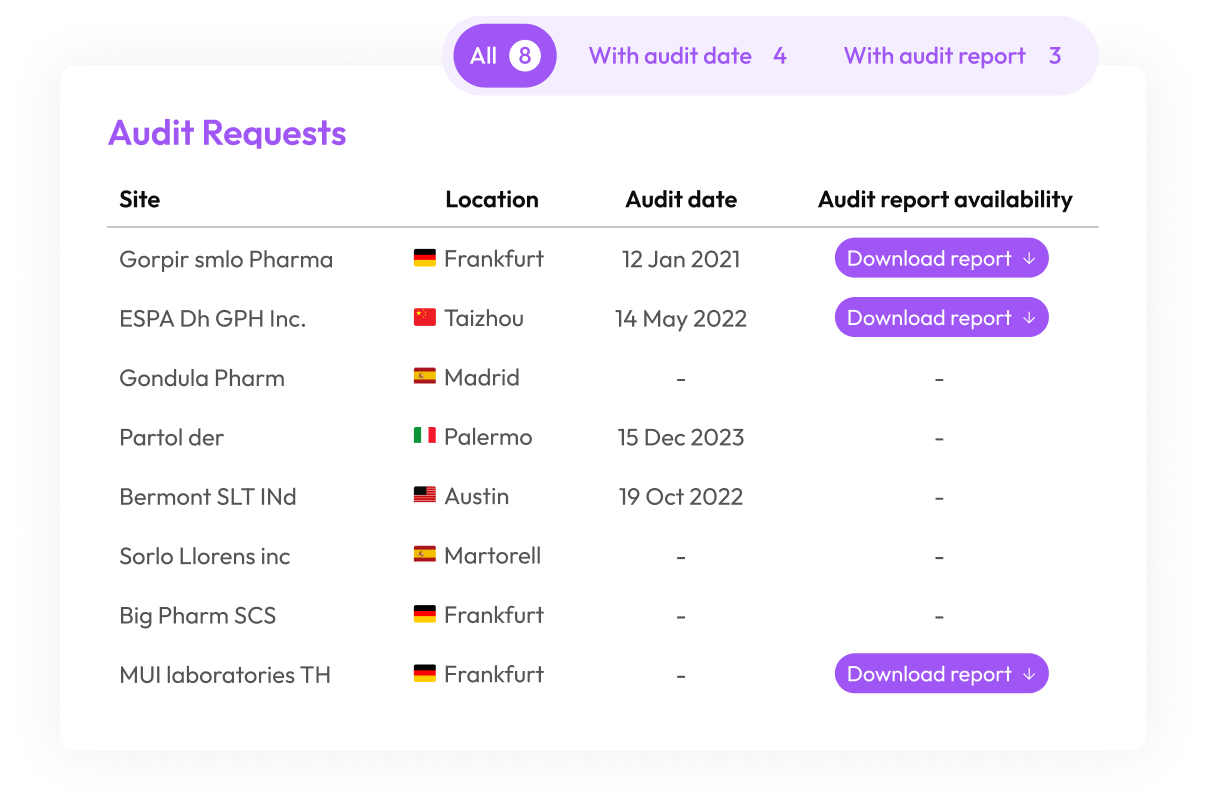
Plan, execute, and track supplier audits globally with a centralized audit management platform built for life sciences supply chains. Access the largest library of GxP audit reports and manage corrective actions with integrated CAPA tracking.
AI-powered platform to schedule, track, and manage internal and third-party audits worldwide.

Access over 5.000+ ready-to-use audits, reducing qualification time by Access over 65% and enabling rapid decision-making.
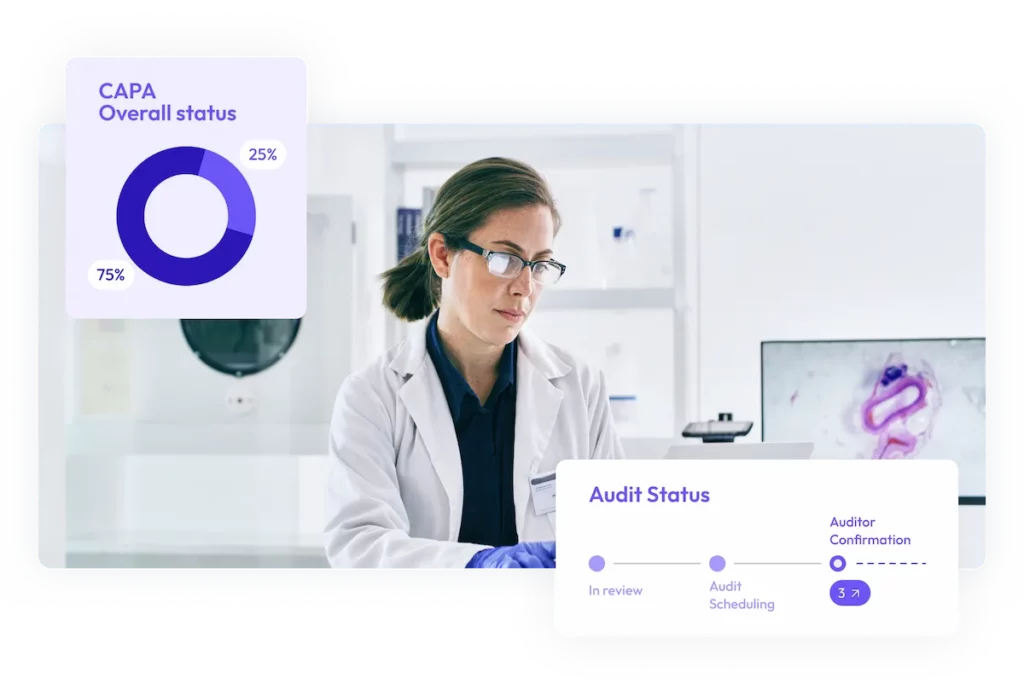
Digitally manage and track CAPAs, monitor progress, and accelerate resolution to ensure continuous compliance.
250+ auditors with local compliance expertise.
Off-the-shelf, shared, or fully customized.
AI-powered audit tracking across your supply chain.
Expertise in 14+ GxP and ISO standards.
Managing a growing global supply network while meeting stringent compliance requirements is no small task, especially with limited internal resources.
By partnering with Qualifyze, Qualix Pharma gained access to a library of over Access over 5.000 audit reports and a scalable solution that accelerated compliance by Access over 65% without increasing operational strain.

Managing supplier audits across a global supply chain requires more than scheduling and documentation. Leading life sciences companies centralize audit planning, maintain continuous visibility into supplier compliance, and ensure corrective actions are tracked through to closure.
A centralized audit platform allows quality and procurement teams to coordinate GxP audits globally, access trusted audit reports, and manage CAPA follow-up across suppliers, while maintaining full visibility into audit performance and supplier compliance.
An audit management platform helps life sciences companies plan, execute, and track supplier audits across their global supply chain. It centralizes audit planning, audit findings, CAPA follow-up, and supplier compliance visibility in one system, enabling quality teams to manage complex GxP audit programs more efficiently.
The Qualifyze Audit Management Platform supports supplier qualification by providing access to the largest library of GxP audit reports and enabling teams to plan and manage supplier audits globally. Quality teams can use audit insights, track CAPA remediation, and maintain visibility into supplier compliance throughout the qualification process.
The platform supports a wide range of supplier audits across the life sciences supply chain, including GMP, GDP, API, excipient, and other GxP-related audits. Companies can coordinate audits across suppliers worldwide and maintain centralized oversight of audit programs.
After an audit is completed, the platform allows quality teams to track corrective and preventive actions (CAPAs), monitor supplier remediation progress, and ensure findings are addressed before closure. This helps maintain compliance and reduces the risk of unresolved audit observations.
Unlike traditional audit tools that only track audit workflows, the Qualifyze platform combines audit management with access to a global library of GxP audit reports. This allows companies to reuse existing audit intelligence, reduce redundant audits, and accelerate supplier qualification.
Audit data provides critical insight into supplier compliance, operational performance, and potential quality risks. By centralizing audit findings and CAPA outcomes, quality teams can identify patterns, monitor supplier performance, and support broader supplier risk management strategies.
Digitize supplier supplier data, simplify compliance, and proactively manage risks.
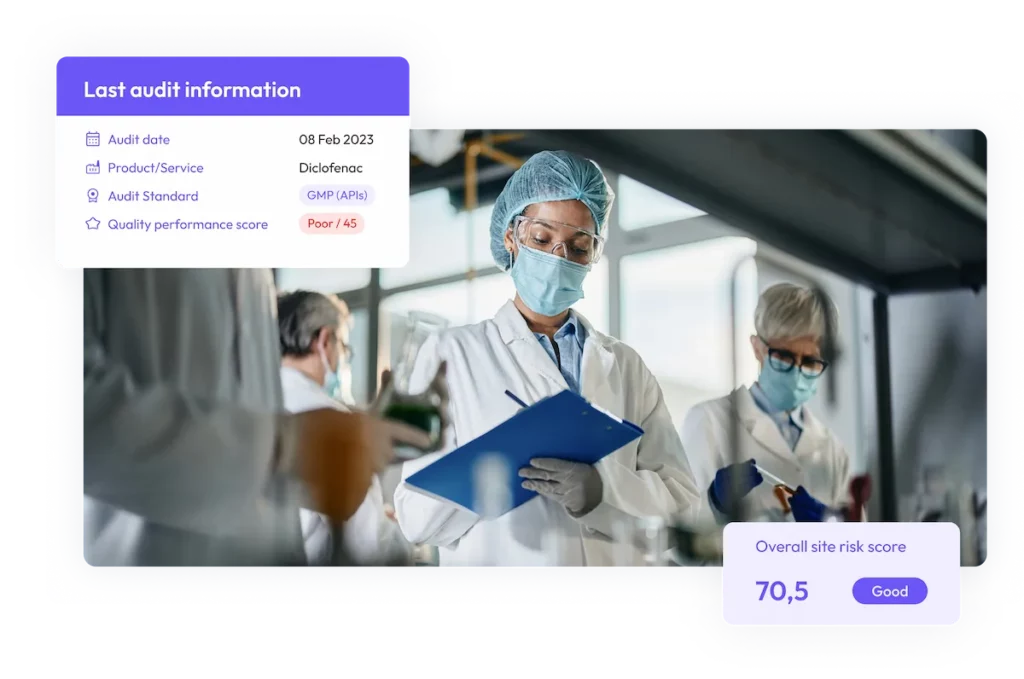
Streamline audits, monitor CAPAs, and access site quality risk assessments in ensure compliance visibility in real-time.
Accelerate supplier selection, minimize sourcing risks, and secure trusted suppliers faster.
