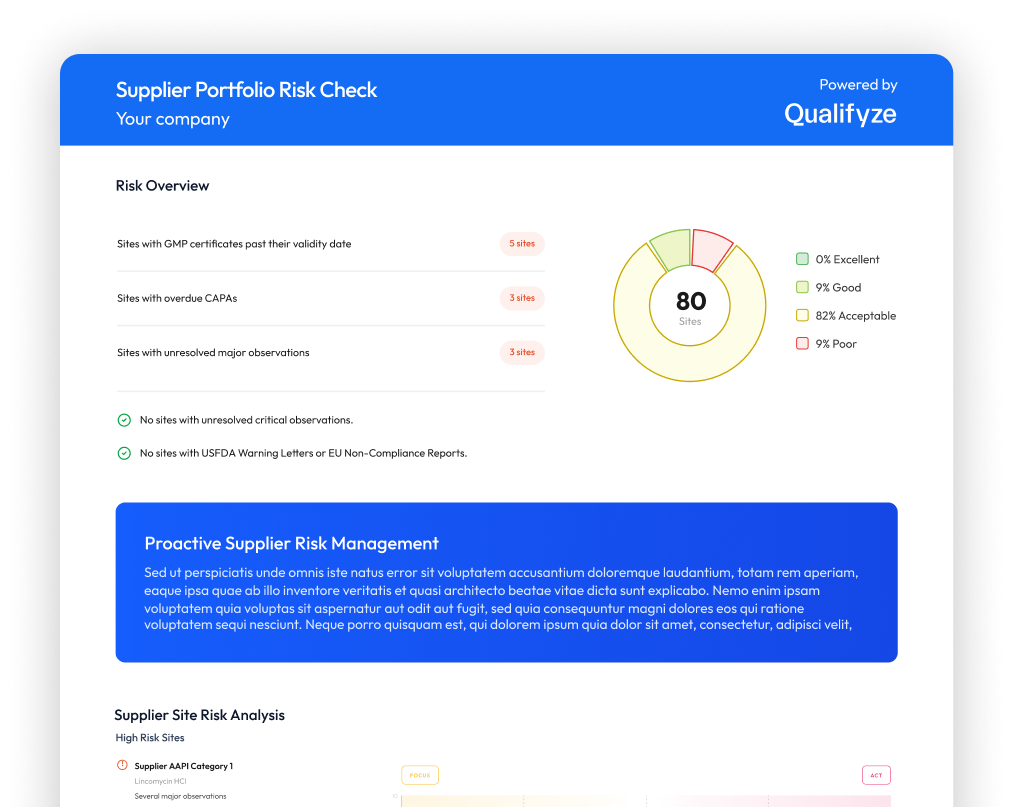
Supplier oversight is typically structured through periodic qualification and monitoring activities, including audits, supplier questionnaires, operational performance metrics, and regulatory or regulatory inspection outcomes.
Each of these mechanisms provides valuable insight into supplier performance, but they are often reviewed separately, at different moments in time, and across different systems.
This approach creates structure, yet industry-level analyses reveal a different reality: supplier risk is not evenly distributed, it does not remain static, and it rarely exists in isolation.
When audit findings, remediation behavior, operational performance signals, and regulatory context are viewed together, clear risk patterns begin to emerge.
The implication is simple: supplier oversight should be driven by real-time supplier risk indicators, not periodic checkpoints.
What Our Analysis Reveals About Supplier Risk
Everyone’s under pressure to cut cost.
But the real opportunity is reducing internal disruptions caused by suppliers.
That’s where dollars are leaking out of the system.
Supply Chain Leader of a Top 5 global pharma company.
Is hidden supplier risk building in your portfolio?
If risk signals concentrate, why is oversight still allocated evenly across suppliers?
Based on aggregated portfolio analysis:
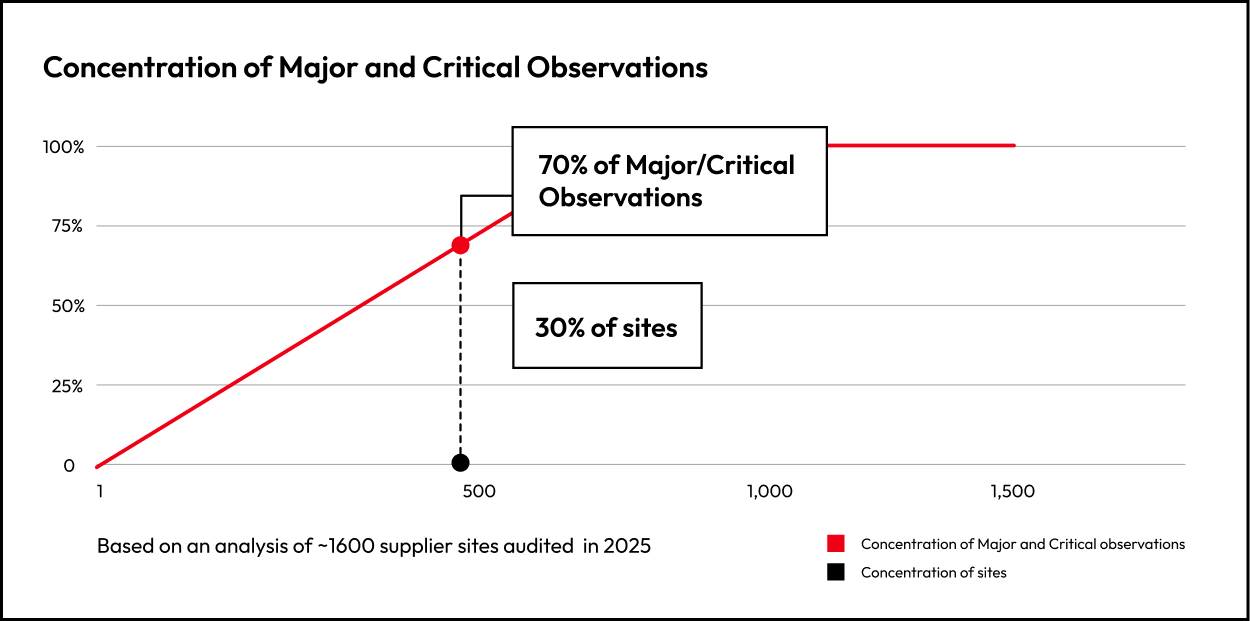
30% of supplier sites drive 70% of total Major and Critical observations. Severity concentrates in a minority of sites.
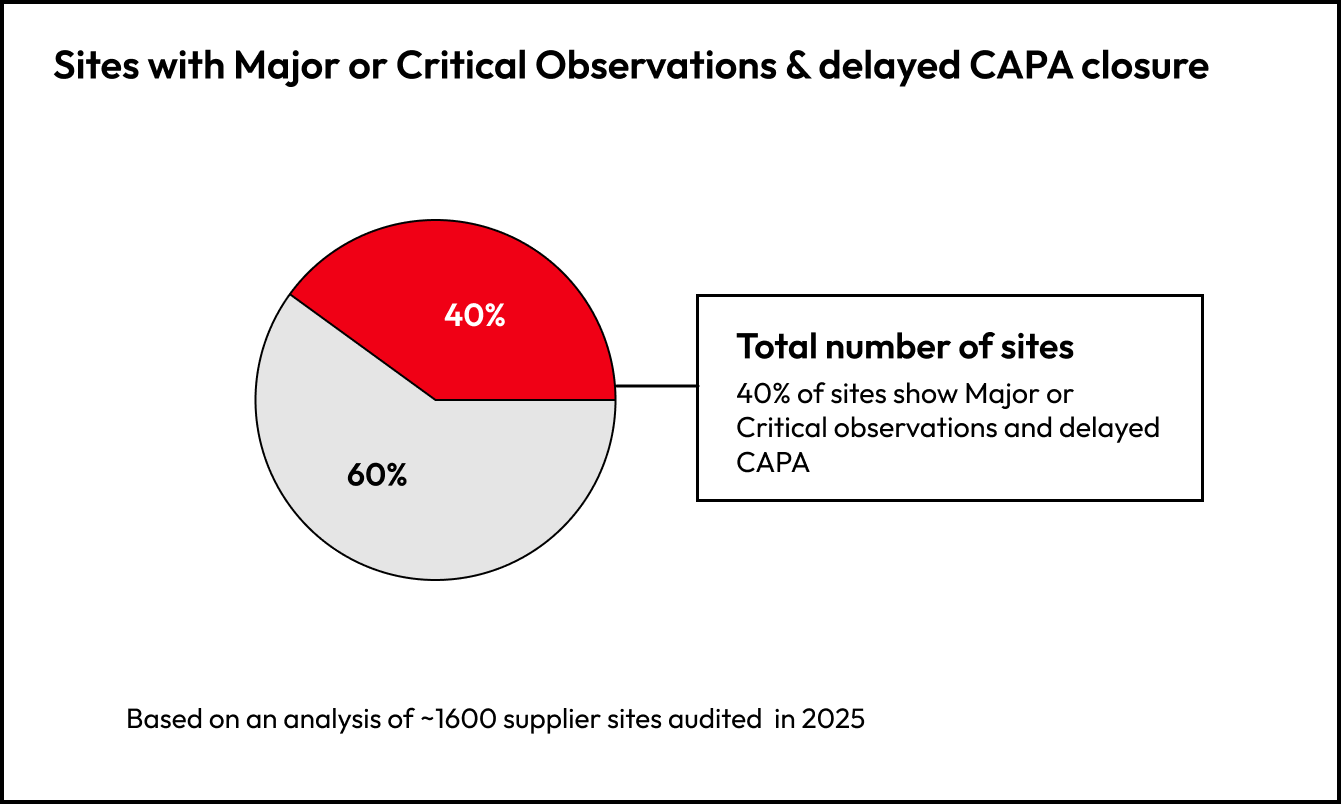
40% of supplier sites show at least one Major/Critical issue or delayed CAPA. Risk signals are not rare, they are present across a meaningful portion of the portfolio.
This distinction matters. Incidences tells us how many sites show issues. Concentration tells us where most risk actually sits.
Yet many supplier oversight programs still rely on fixed monitoring cycles and periodic checkpoints.The tension becomes visible:
Risk signals are measurable and concentrated, but oversight is often distributed evenly over time. When exposure clusters in a subset of sites, uniform monitoring spreads attention thinly and may underweight where severity actually accumulates.
Periodic assessments capture supplier quality at a moment in time. What determines real risk is what happens afterward.
Across aggregated and anonymized portfolio analysis:
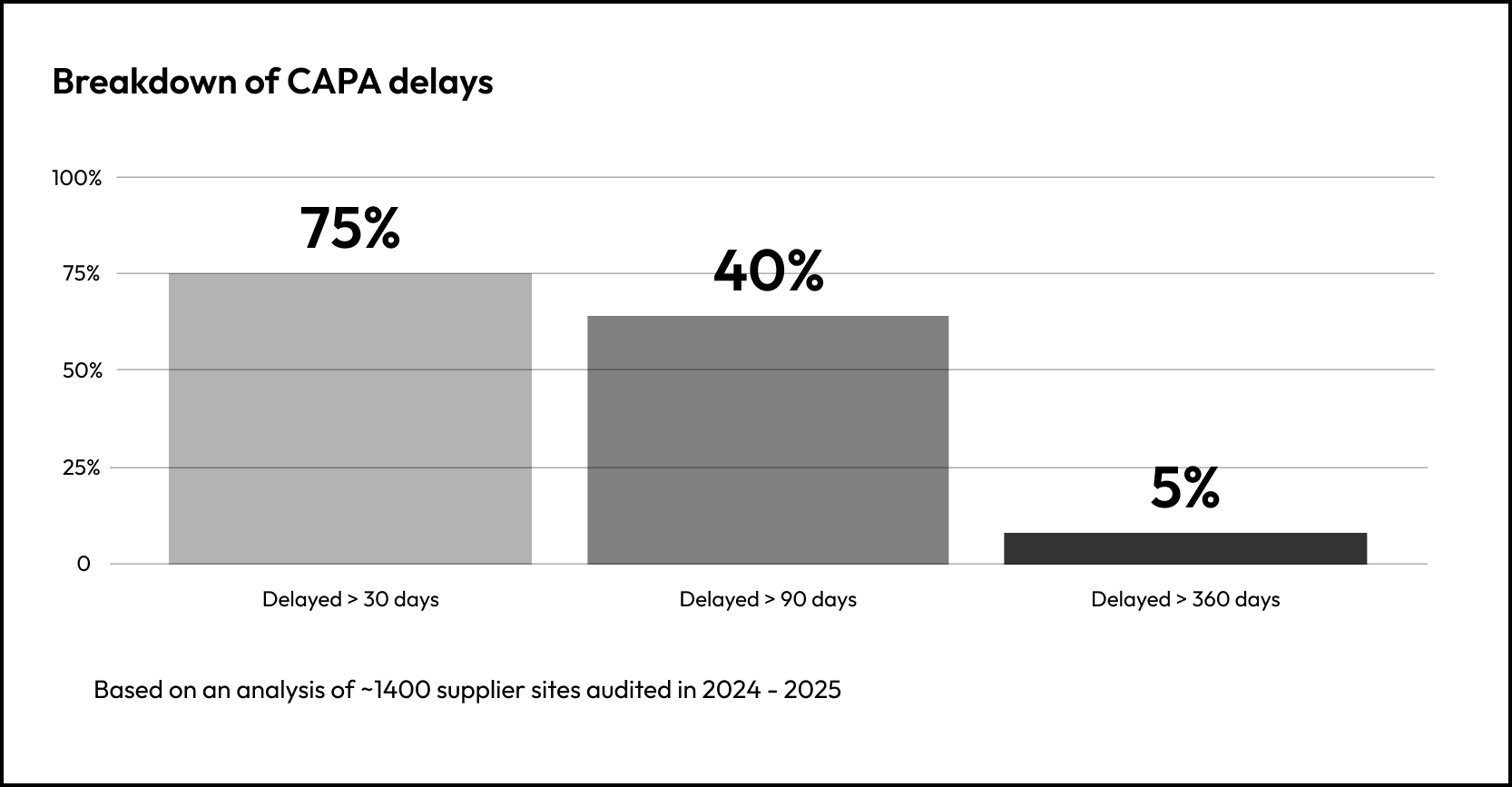
Two suppliers may share the same audit conclusion. Their residual risk after a few months will likely differ materially.
If you want to understand supplier site risk, don’t ask “Did the site pass?”… Ask “How long does risk remain open and are they remediated on-time?”
Supplier risk does not stop at the initial assessment. It continues to evolve as performance signals accumulate.
Risk exposure is shaped by multiple factors – remediation behavior, operational performance trends, supervisory posture, and broader supply chain context.
When these signals are evaluated together:
Within 12 months, only 16% of suppliers show genuine improvement. Performance stabilizes, remediation happens on time, and residual risk reduces. The remaining 84% show measurable deterioration, with remediation delays, operational performance drift, and emerging supervisory pressure.
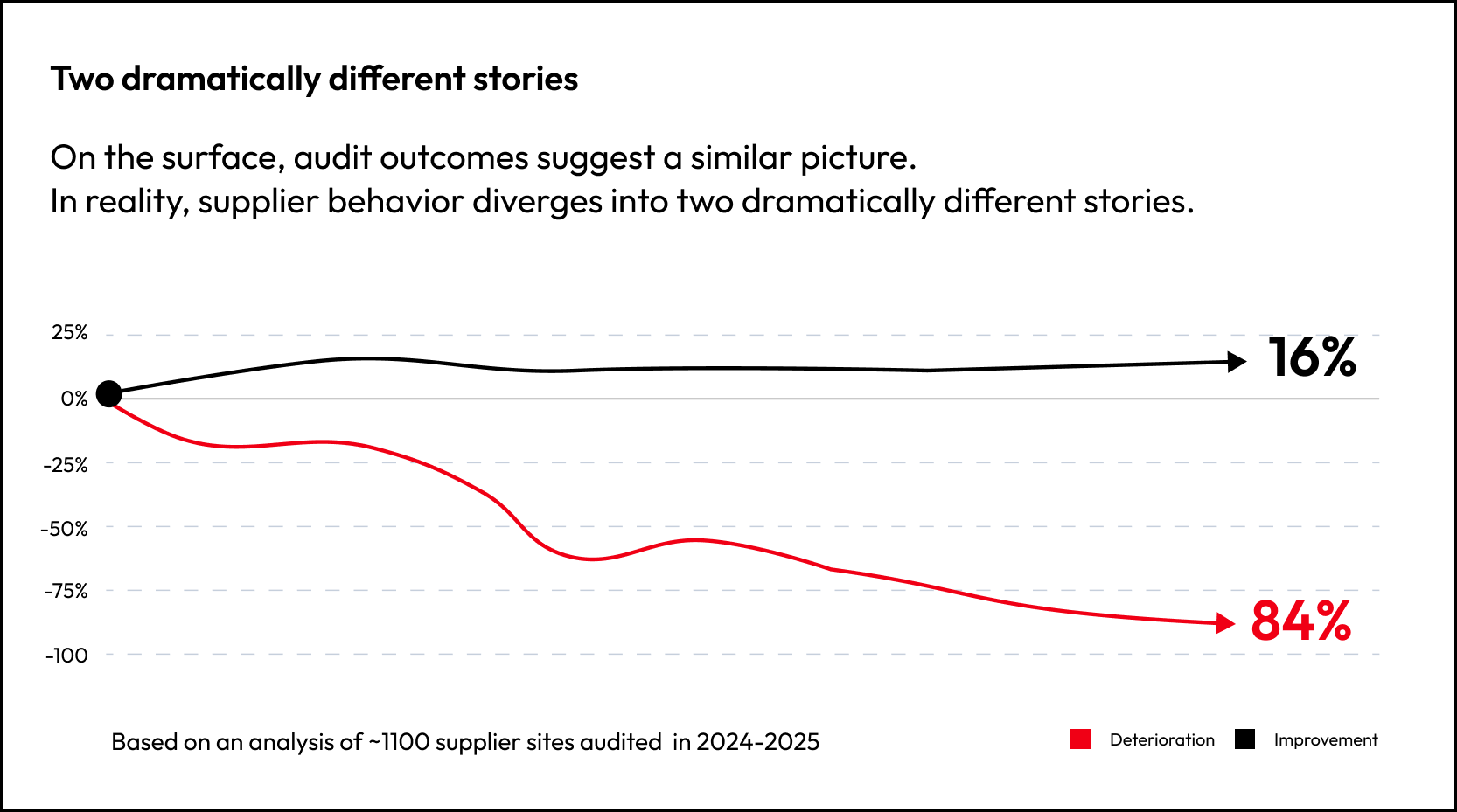
Importantly, this movement often occurs without any change in the original assessment outcome. The initial review captures a point in time. The risk profile evolves afterward.
Without integrating signals continuously across systems, this shift in risk exposure can remain partially invisible.
Supplier Type Sets the Baseline. Behavior Determines the Risk.
At first glance, supplier performance across segments looks relatively similar.
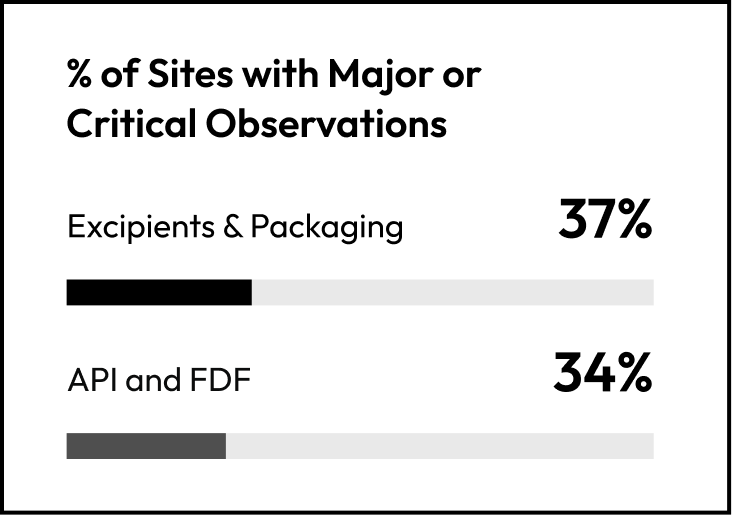
On observation severity alone, no segment appears structurally weaker than the others. But severity at audit is only the starting point. When remediation behavior is layered in (delayed CAPAs) a different pattern emerges.
35% of Excipients sites show both elevated severity and delayed remediation. That compares to roughly 25% of FDF and API sites, and 16% of packaging sites.
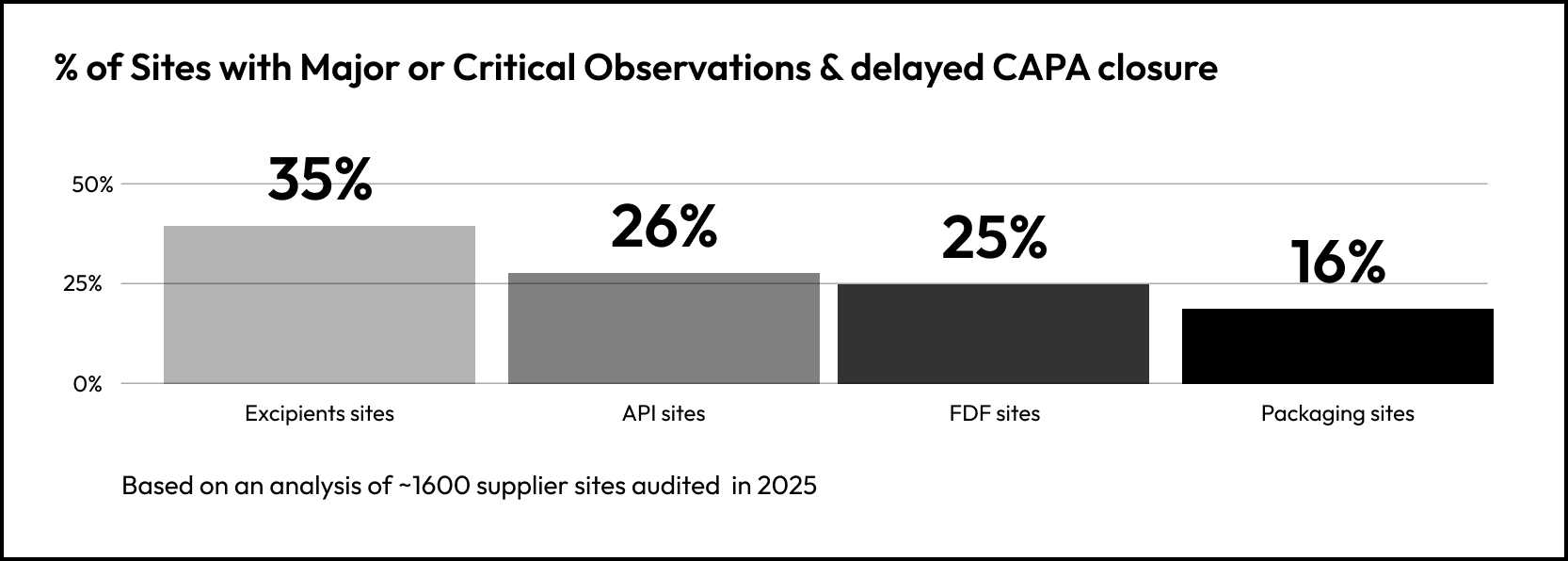
The difference is not in observation severity, it is what happens after the audit.
API suppliers often respond faster, likely reflecting higher regulatory scrutiny and commercial pressure.
Packaging issues are typically more contained and operationally simpler to resolve.
Excipient sites, by contrast, show more persistent remediation delays, meaning risk exposure remains active longer.
Treating segments uniformly may overlook where exposure persists, not because findings were more severe, but because closure takes longer. That is where risk begins to diverge.
Why do these patterns remain difficult to see?
Because supplier risk signals are fragmented.
Additional risks, such as ESG, Financial Risks or geopolitical risks are different systems and departments entirely.

Teams spend significant time gathering and reconciling data instead of interpreting it. Industry research suggests quality professionals spend over 30% of their time stitching information together across systems.¹
Meanwhile, regulators continue to identify concentrated quality deterioration in specific manufacturing segments.²
The issue is not a lack of effort. It’s that no single view brings these signals together in a way that clearly shows where risk actually sits.
- Oversight defaults to even distribution.
- Residual risk exposure remains embedded.
- Behavioral drift goes undetected.
- Allocation aligns with risk intensity.
- Remediation durability becomes comparable.
- Portfolio risk becomes segmentable.
Supplier oversight shifts from periodic validation to risk intelligence.
Test These Patterns Against Your Portfolio
If risk is concentrated, evolving, and structurally clustered, the key question becomes:
Is this happening in your portfolio?
Risk concentration cannot be understood through static reports or isolated metrics. It requires viewing audit outcomes, remediation behavior, and operational and regulatory inspection outcomes together.
Unlock Your Complimentary Supplier Base Risk Check
To understand whether these risk patterns exist in your supplier base, request a focused Supplier Base Risk Check.