From Compliance Gaps to AI-Powered Quality Managementt
Centralize quality data, predict risks early, and stay ahead of compliance issues, before they escalate.
How Qualifyze solves your challenges

Unified Quality Data Hub
Leverage Qualifyze AI capabilities to unify audit reports, supplier data, and regulatory insights, streamlining workflows and ensuring greater accuracy.

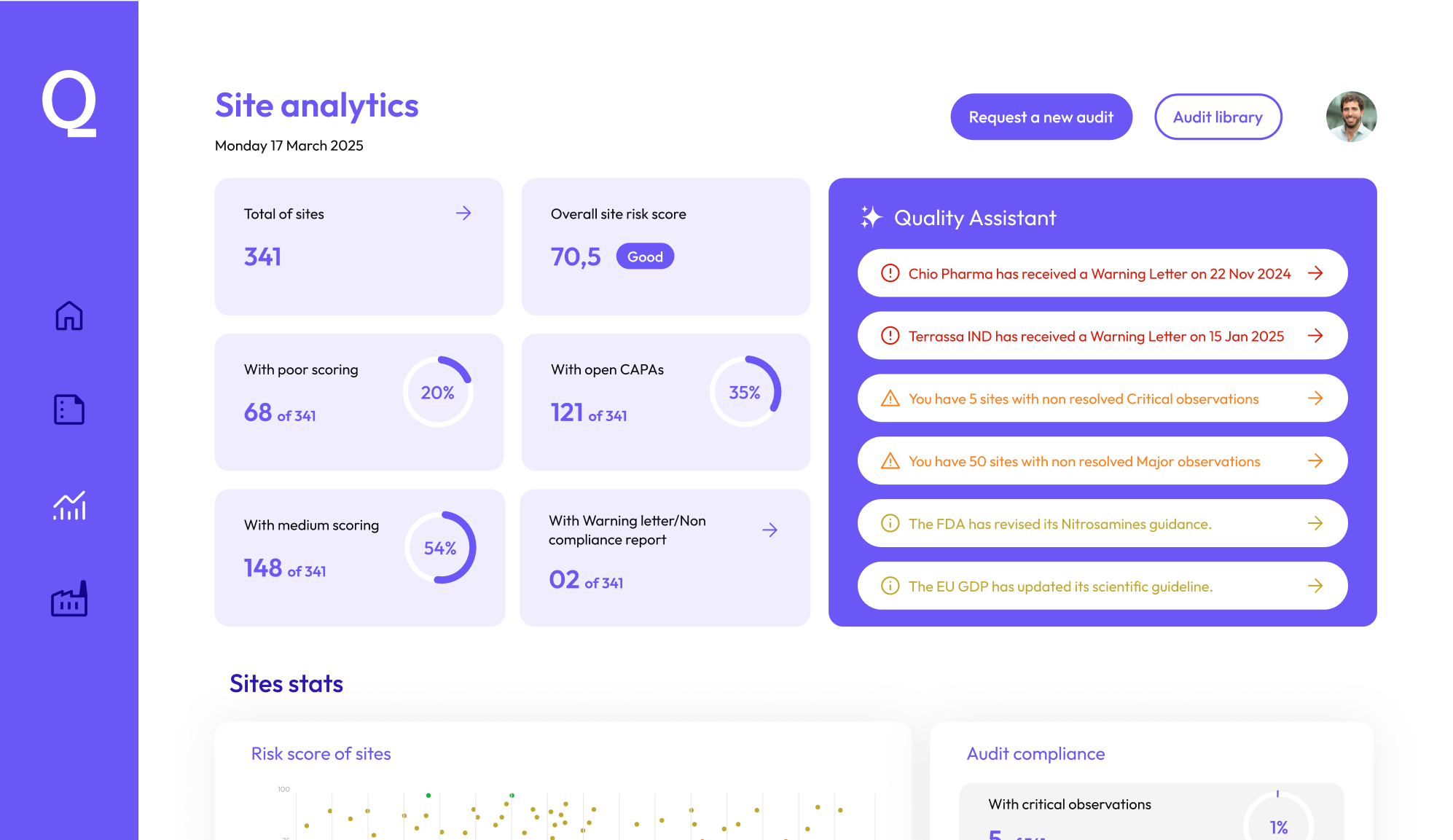
Efficient and effective Quality Risk Management
Smart resource allocation powered by AI. Prioritize risks and equip auditors with global market insights for stronger preparation.

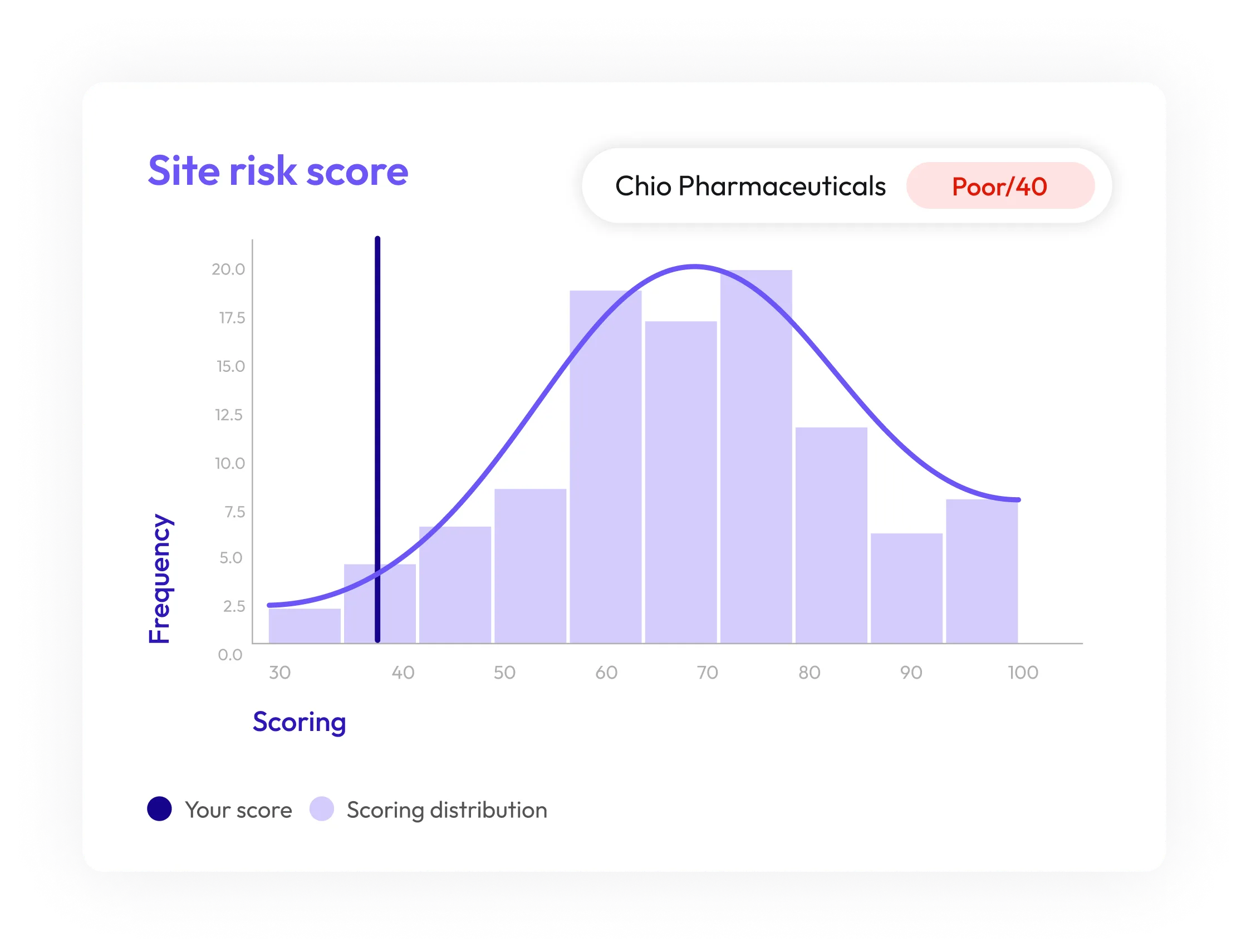
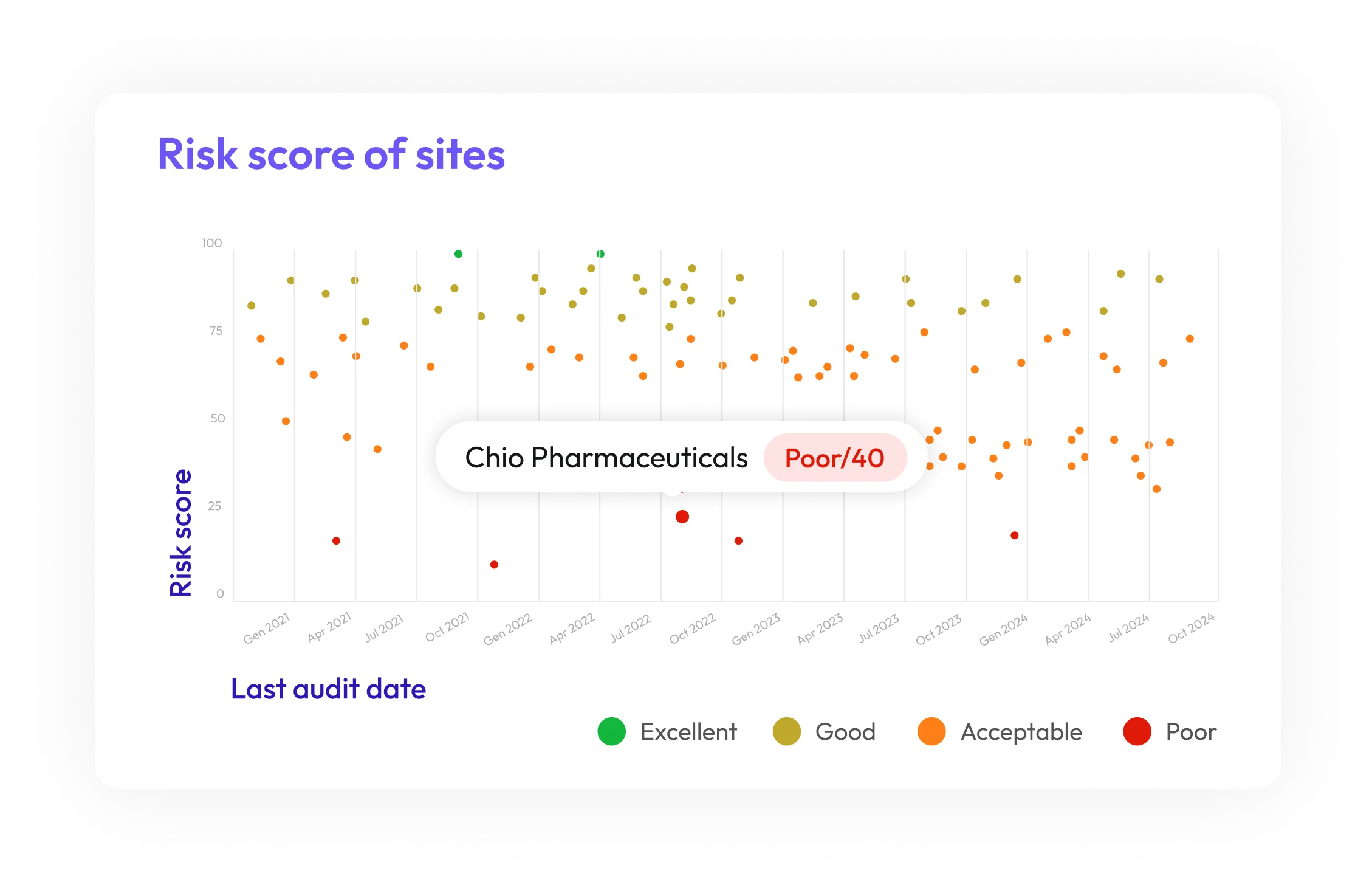
Proactive Quality Risk Identification
Spot potential supplier compliance issues before they escalate, using predictive analytics to stay ahead of risks and maintain uninterrupted operations.

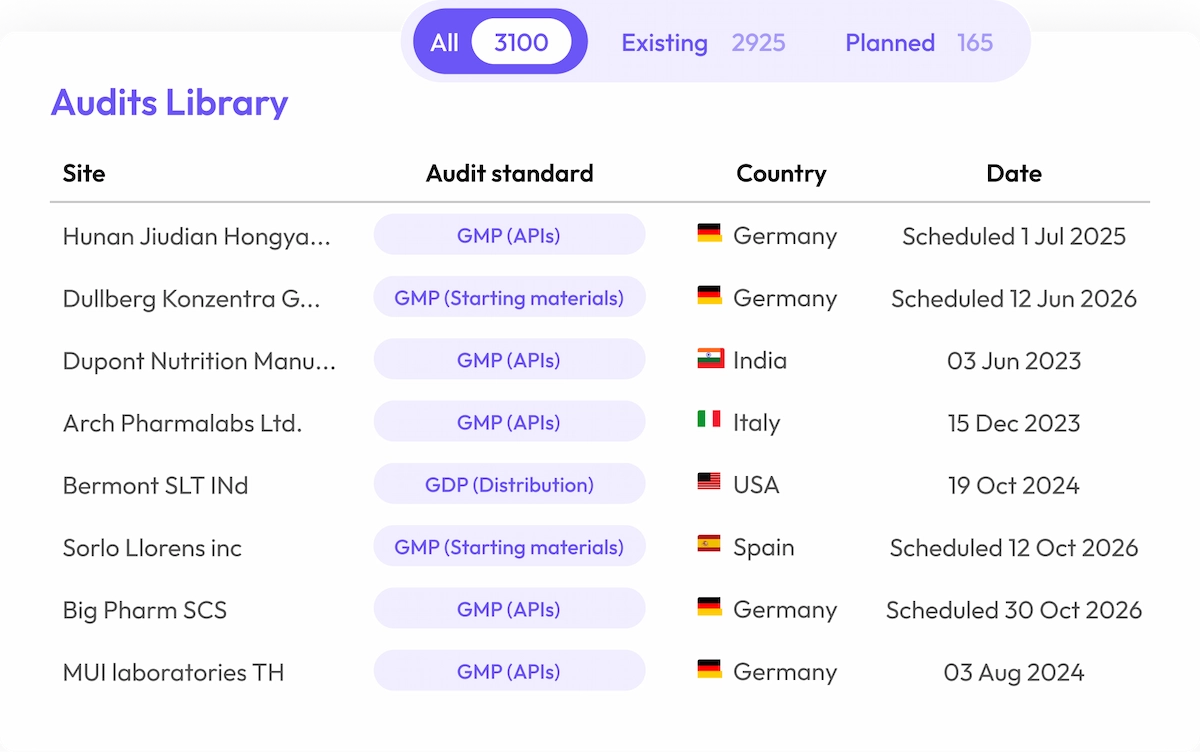
Accelerate Supplier Compliance
Gain instant access to over 5.000 audit reports, speeding up supplier qualification by 65%, saving valuable internal time and resources.

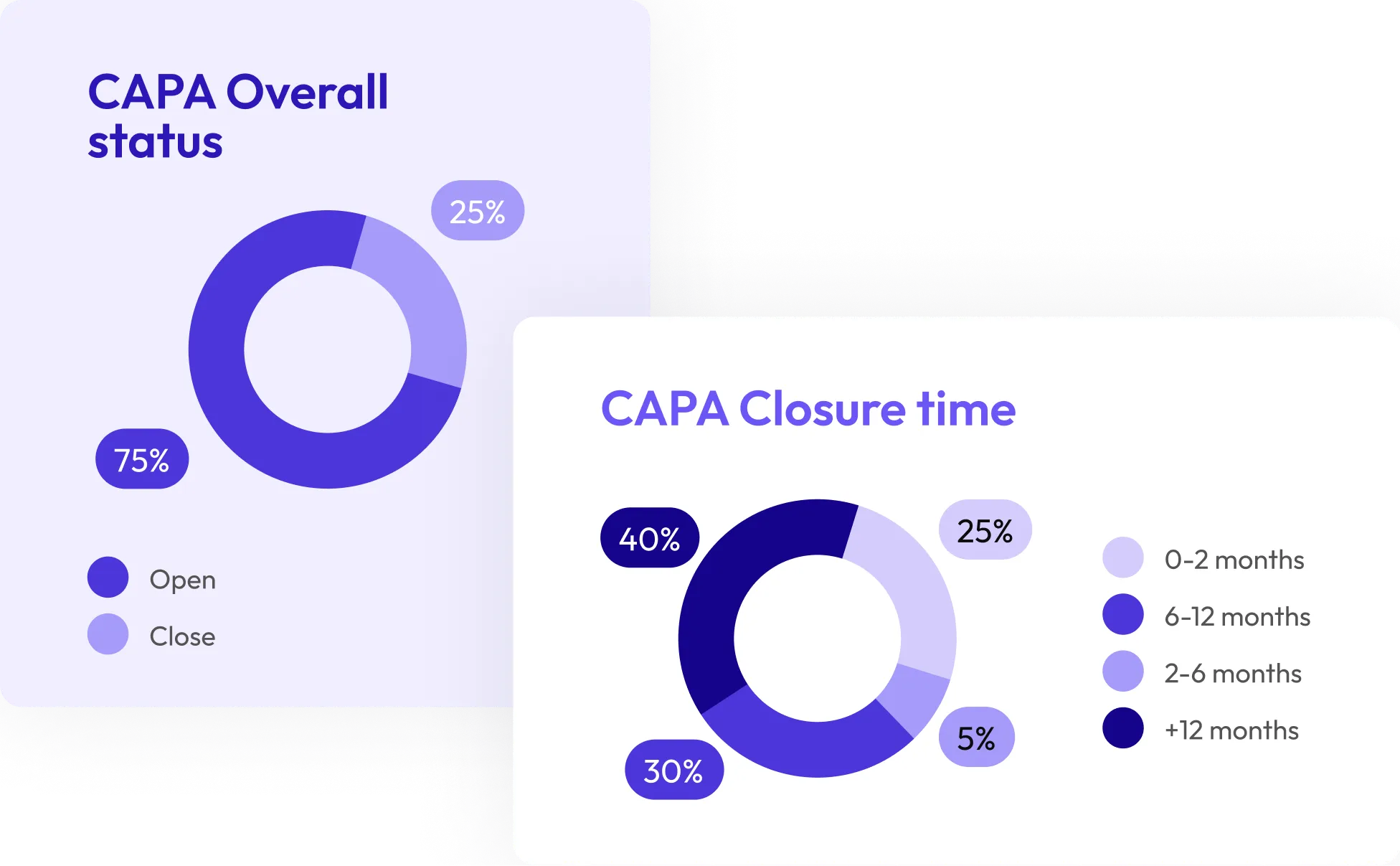
Simplified CAPA Management
Digitally manage CAPAs while AI benchmarks supplier performance, ensuring timely closures and driving continuous quality improvement.

Proven outcomes for Quality teams
Shift from reactive to proactive supplier risk management
Real-time view of your supplier performance
Benchmark your supplier compliance
Explore the tools driving Proactive Compliance and Supplier Risk Management
Audit Management Platform
Centralize and simplify global audit management. Plan, execute and track audits with AI, so your team can focus on what really matters.
Quality Insights Platform
Turn disconnected compliance data into AI-powered insights. Monitor supplier performance in real time, detect risks early, and drive smarter, faster decisions.
What they say about us?
”It’s the closest to having an extra member in our team. From alerts to CAPA follow-ups to structured reports, Qualifyze gives us everything we need while making the most out of our resources.”
“We chose to collaborate with Qualifyze, not only for their extensive audit database and fast delivery times but also for the precision and high quality of their reports.”
“Aenova is aware of this responsibility and therefore prefers to work with excellent partners, such as Qualifyze”
“Qualifyze helped us reduce costs and time thanks to their team of experienced auditors.”
“When Qualifyze audited us in 2021, we were impressed by the auditor’s qualification, precision, and profound knowledge. The audit also helped us a lot since it was comparable to an authority audit.”
“We have valued our experience working with Qualifyze on audit reports because of their reliable customer service, collaboration and thoroughness.”
“We chose Qualifyze because of their reliable customer service and the good quality of the audit reports at a fair price. Our requests were handled with flexibility and willingness to compromise.”