Predict and proactively manage supplier risks with AI-powered insights
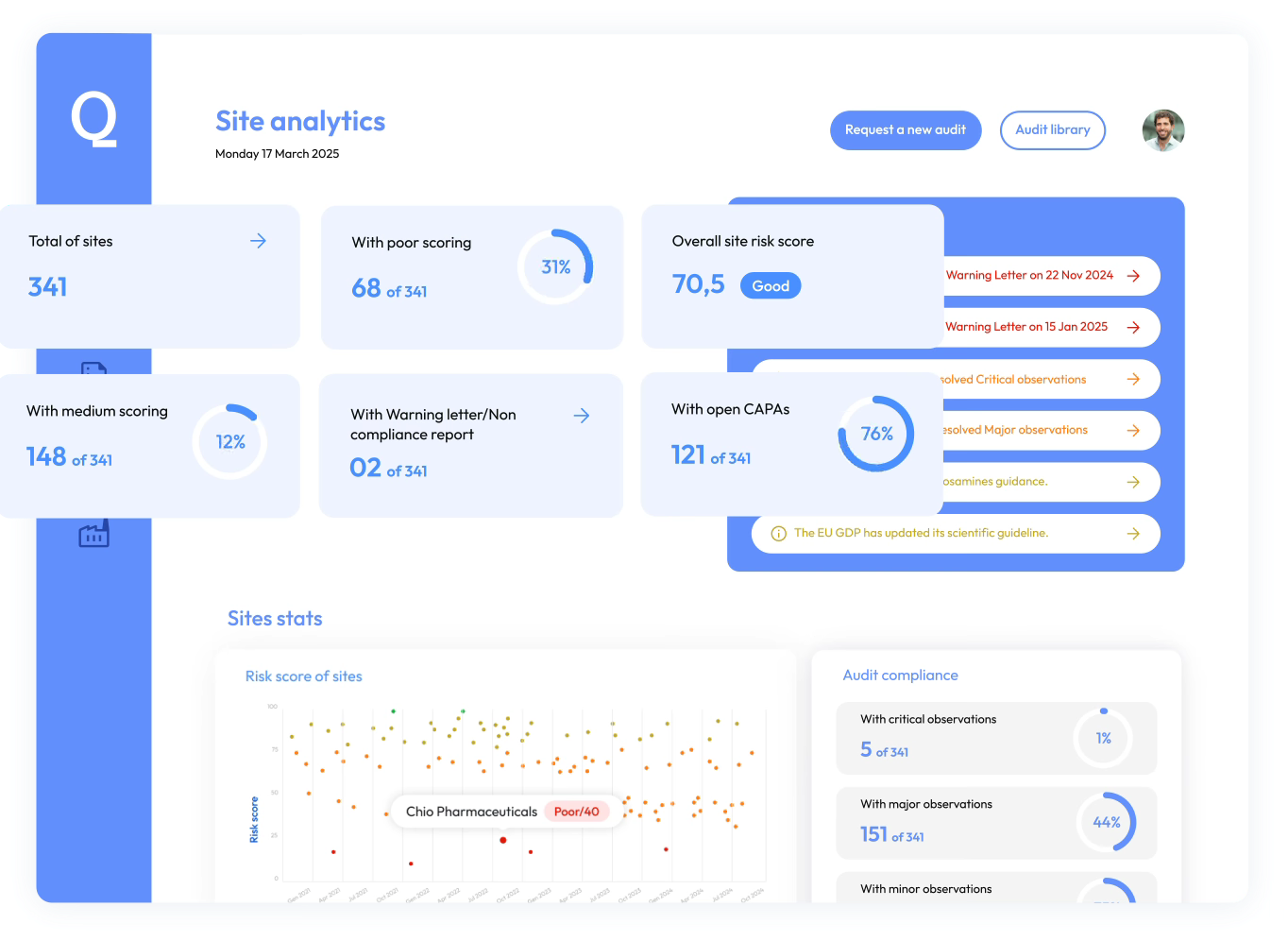
Identify, monitor, and mitigate supplier risk continuously across your global supply chain using AI-driven insights built on audit outcomes, operational quality metrics, inherent manufacturing risk, and regulatory supervision signals.
Access a unified view of supplier risk by combining audit outcomes, operational performance indicators, and regulatory supervision signals

Prioritize high-risk suppliers and cut down on unnecessary audits to make your compliance process more efficient.

Use supplier risk insights to prioritize qualification decisions, identify high-risk suppliers earlier, and maintain continuous oversight across your supplier network.
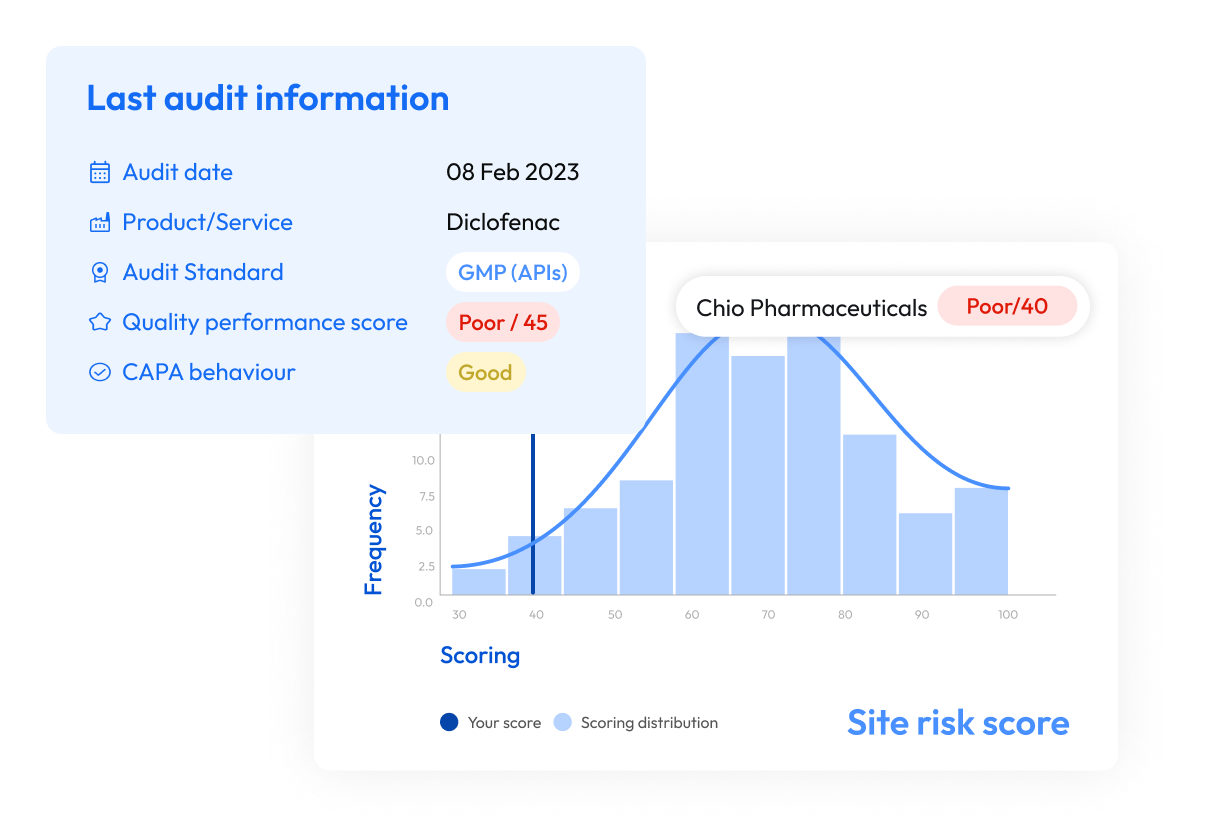
Unify compliance and sourcing data to improve supplier selection, qualification, and long-term risk management.
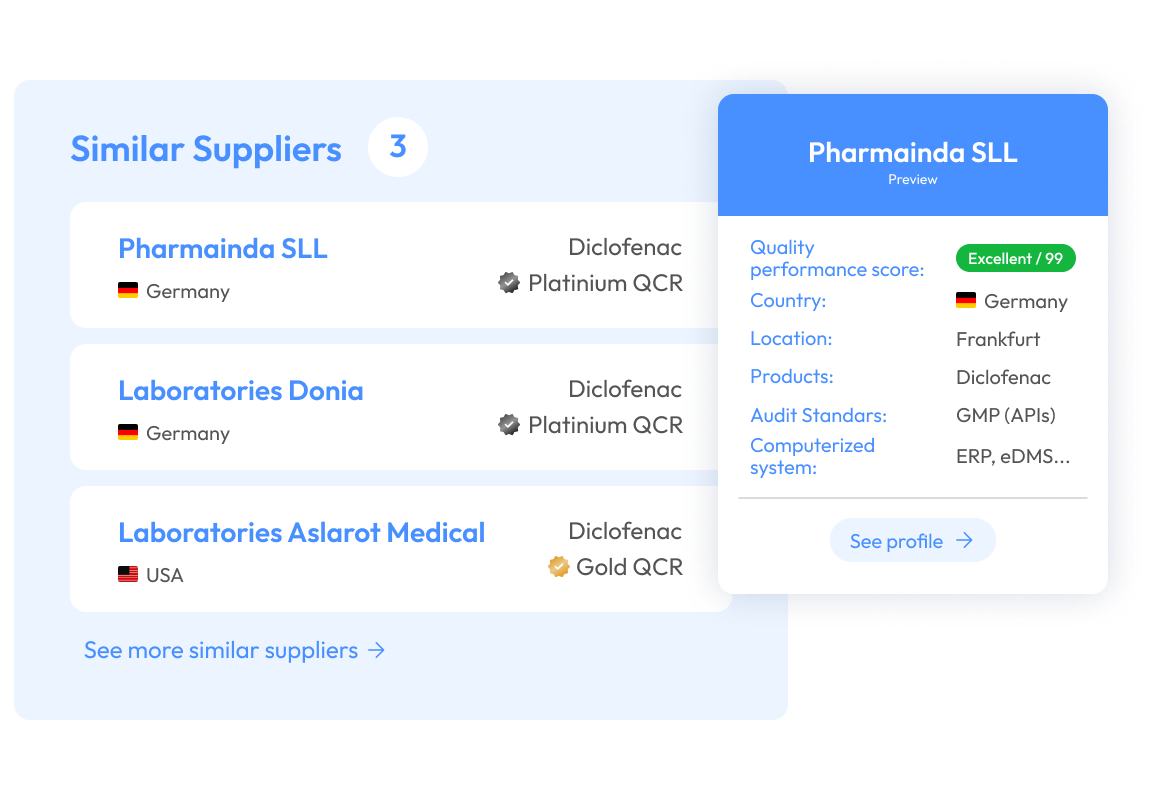
All your supplier risk and compliance data in one place.
AI-powered insights help you evaluate supplier risk in seconds.
Prioritize the suppliers that need your attention most.
Align quality and procurement teams through data.
Managing supplier risk across a global life sciences supply chain requires more than periodic audits. Quality teams must continuously monitor supplier performance, evaluate regulatory signals, and assess operational risk factors that could impact product quality or compliance.
Supplier risk management platforms help organizations centralize supplier intelligence, identify risk trends earlier, and support supplier qualification decisions using data from audits, operational performance, and regulatory oversight.
The Quality Insights Platform evaluates supplier risk using a multi-factor model that combines audit outcomes, operational quality metrics, inherent manufacturing risk, and regulatory supervision signals such as inspections and warning letters.
Continuous supplier risk monitoring allows quality teams to prioritize supplier qualification efforts based on real-time risk insights. By combining audit data with operational and regulatory intelligence, teams can identify high-risk suppliers earlier and make more informed qualification decisions.
Supplier risk evaluation incorporates multiple signals including audit observations, CAPA performance, operational quality indicators, regulatory inspections, and supervisory actions from authorities such as the FDA or EMA. It also considers inherent risk factors such as the complexity of the technologies used at a supplier and the geographic risk associated with the location of the manufacturing supplier.
Supplier audits provide point-in-time assessments of supplier compliance. Supplier risk management platforms provide continuous oversight by combining audit results with operational and regulatory signals to monitor supplier performance over time.
Digitize supplier data, simplify compliance, and proactively manage risks.
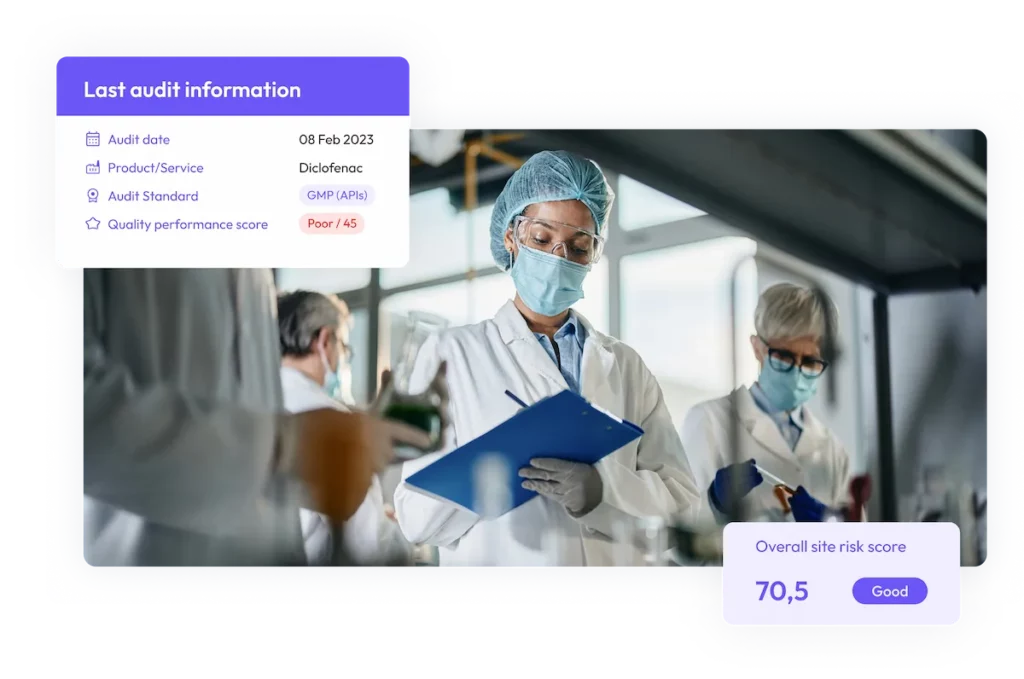
Streamline audits, monitor CAPAs, and access site quality risk assessments in ensure compliance visibility in real-time.
Accelerate supplier selection, minimize sourcing risks, and secure trusted suppliers faster.
